April 2025
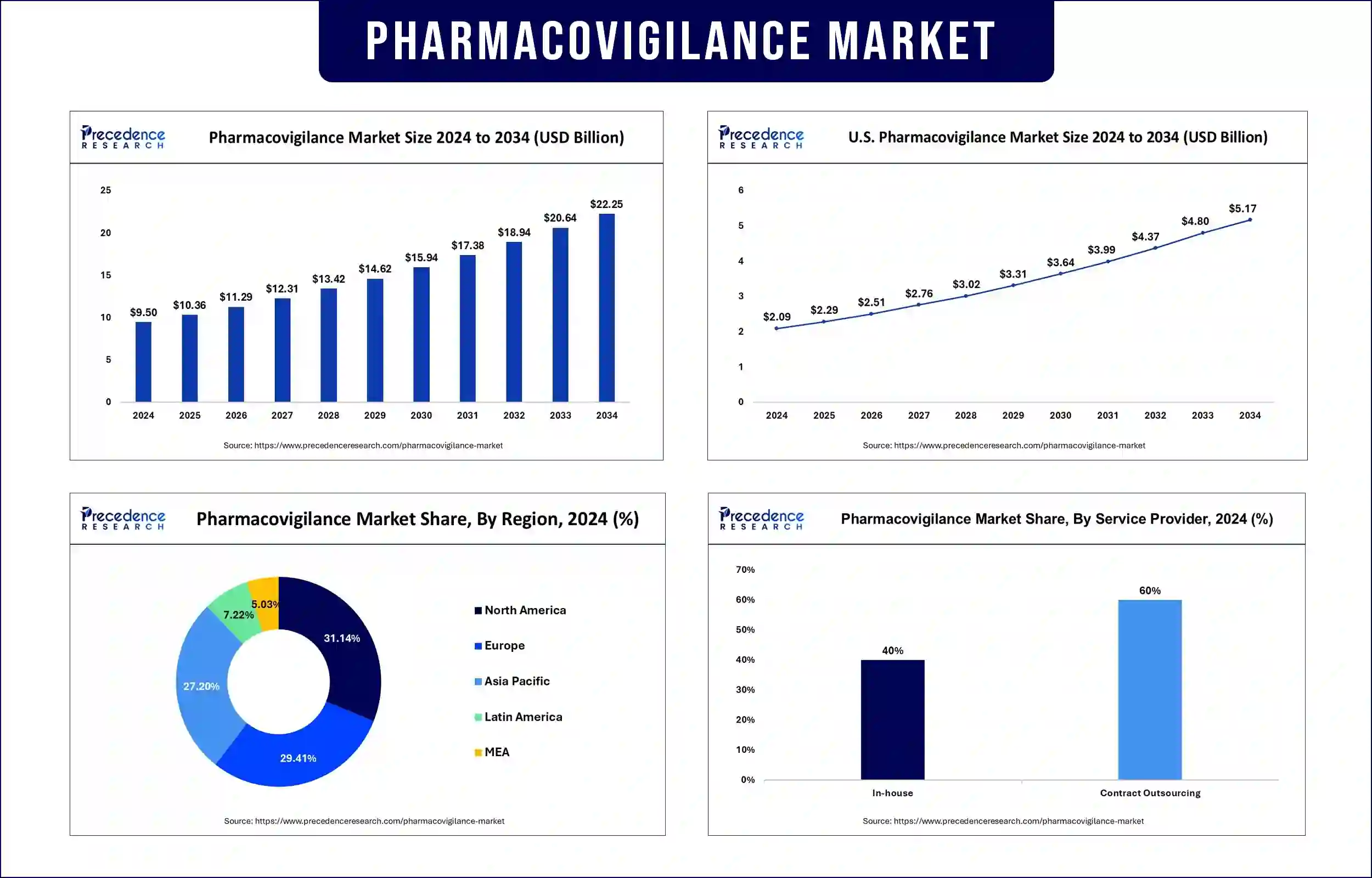
The global pharmacovigilance market revenue reached USD 10.36 billion in 2025 and is predicted to attain around USD 20.64 billion by 2033 with a CAGR of 8.88%. Expanding rapidly due to the increasing focus on drug safety, stringent regulatory requirements, and the rising incidence of adverse drug reactions (ADRs).
The pharmacovigilance market encompasses a wide range of activities, including case reporting, risk management, and regulatory compliance, aimed at enhancing drug safety. Pharmaceutical companies, contract research organizations (CROs), and regulatory agencies are increasingly investing in advanced data analytics and real-world evidence (RWE) to improve pharmacovigilance outcomes. Advancements in chronic diseases with the parallel growth of global drug development led to an increased urgency for predictive adverse event monitoring.
The WHO Programme for International Drug Monitoring and the FDA’s Sentinel Initiative work together to speed up drug-related risk detection and prevention efforts. The FDA started the Sentinel Initiative in 2008, and it developed into a complete system to track the safety aspects of FDA-regulated products. Enhanced pharmacovigilance practices receive a commitment from public health stakeholders worldwide through their implemented global efforts.
Growing Regulatory Focus on Drug Safety
Global regulatory agencies, including the FDA, EMA, and WHO, are enforcing stricter pharmacovigilance guidelines to mitigate drug safety risks. The FDA’s Adverse Event Reporting System (FAERS) and WHO’s VigiBase database have implemented expanded abilities to detect adverse events. Drug safety detections occur earlier through these advancements while regulatory responses become faster, thus minimizing patient exposure to risks and ensuring effective drug performance.
Increasing Adoption of AI in Drug Safety Monitoring
AI-powered pharmacovigilance tools are improving real-time case assessment, reducing manual workload, and enhancing accuracy in adverse event reporting. The pharmacovigilance operations become automated through AI technologies. The systems reduce expenses while improving adherence to regulatory standards. The U.S. Food and Drug Administration (FDA) identified an acute rise of artificial intelligence and machine learning (ML) applications within Investigational New Drug (IND) processes across different fields that include pharmacovigilance.
Expansion of Pharmacovigilance Outsourcing Services
Companies in the pharmacovigilance market are increasingly outsourcing drug safety activities to CROs and specialized pharmacovigilance service providers. Operating from this strategic move enables companies to decrease their operational expenses without breaching global pharmacovigilance regulatory standards. The World Health Organization (WHO) maintains that proper pharmacovigilance systems must exist because they secure patient safety effectively. The use of external expertise enables pharmaceutical companies to handle core competencies better while speeding up new therapy development and sustaining strict safety tests against complex regulations.
Rising Importance of Real-World Data (RWD) and Real-World Evidence (RWE)
Regulatory agencies and pharmaceutical companies are leveraging RWD and RWE to enhance drug safety assessments, identify long-term risks, and improve patient outcomes. The worldwide move toward medical product assurance includes RWD along with RWE in regulatory decisions. The World Health Organization (WHO) recognizes RWE's capability to strengthen pharmacovigilance practices, thus supporting its implementation into the drug safety framework. Public health benefit arises from stakeholders who adopt RWE and RWD approaches to drug safety management.
North America is projected to dominate the pharmacovigilance market, driven by stringent FDA guidelines, rising drug approvals, and increasing adoption of automated pharmacovigilance solutions. Strong pharmacovigilance practices have become essential to monitor drug safety during the post-market phase, as recent increases in drug approvals. The FDA's guidance under Good Pharmacovigilance Practices and Pharmacoepidemiologic Assessment requires both safety signal identification and complete pharmacovigilance plan development.
Automation in pharmacovigilance systems now enables immediate adverse event monitoring together with regulatory obligations compliance. These important factors have established North America as the top market leader in pharmacovigilance, which secures both the effectiveness and safety of treatment medications.
Asia Pacific is anticipated to experience the fastest growth in the pharmacovigilance market, attributed to increasing clinical trial activities, government-led pharmacovigilance programs, and the rising presence of CROs in countries like India and China. Chinese clinical research falls under the National Medical Products Administration (NMPA), which uses streamlined regulations to develop pharmaceutical innovation.
Healthcare experts recommend that India reform its clinical trial regulations to enhance market participation in the worldwide sector. The region shows its dedication to drug safety and efficacy enhancement through strong pharmacovigilance practice development. Regulatory agencies in emerging economies are strengthening pharmacovigilance frameworks, encouraging pharmaceutical companies to enhance their drug monitoring systems.
| Report Attribute | Key Statistics |
| Market Revenue in 2025 | USD 10.36 Billion |
| Market Revenue by 2033 | USD 20.64 Billion |
| CAGR | 8.88% |
| Quantitative Units | Revenue in USD million/billion, Volume in units |
| Largest Market | North America |
| Base Year | 2024 |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
By Clinical Trial Phase
By Service Provider
By End User
By Therapeutic Area
By Type
By Process Flow
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape
overview @ https://www.precedenceresearch.com/sample/1442
You can place an order or ask any questions, please feel free to contact at sales@precedenceresearch.com | +1 804 441 9344
April 2025
January 2025
April 2025
January 2025