What is Clinical Data Management Market Size?
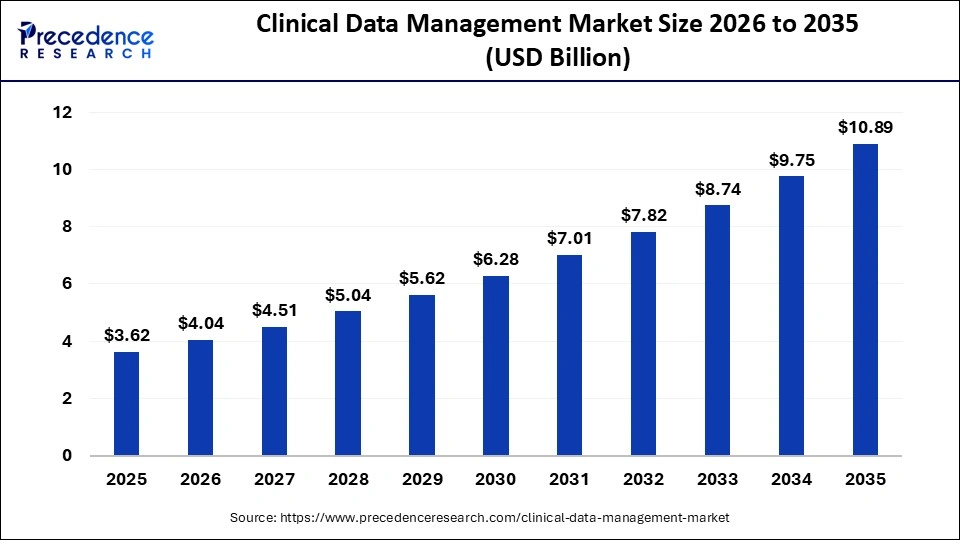
The global clinical data management market size is calculated at USD 3.62 billion in 2025 and is predicted to increase from USD 4.04 billion in 2026 to approximately USD 10.89 billion by 2035, expanding at a CAGR of 11.64% from 2026 to 2035. This market is growing as digital platforms are increasingly adopted to streamline data collection, analysis, and regulatory compliance in clinical trials.
Market Highlights
- North America dominated the market, having the biggest market share of 43.40% in 2025.
- The Asia Pacific is expected to grow at a notable CAGR of 12.5% between 2026 and 2035.
- By component, the services segment contributed the highest market share of 68.50% from 2026 to 2035.
- By component, the software segment is growing at a solid CAGR of 11.2% between 2026 and 2035.
- By delivery mode, the web-based/cloud-based segment generated the largest market share of 62.60% in 2025.
- By delivery mode, the on-premise segment is expanding at a healthy CAGR of 11.1% between 2026 and 2035.
- By tool/platform, the EDC systems segment captured the biggest market share of 43.3% in 2025.
- By tool/platform, the ECOA/ePRO segment is projected to grow at a strong CAGR of 10.9% between 2026 and 2035.
- By end-user, the pharmaceutical and biotechnology companies segment recorded the biggest market share of 42.4% in 2025.
- By end-user, the CROs segment will grow at a notable CAGR of 11.5% between 2026 and 2035.
Market Overview
Is Smarter Data the Secret Fuel Behind Faster Drug Approvals?
The clinical data management market is experiencing steady growth, driven by the need for faster, more accurate decision-making as the volume and complexity of clinical trial data continue to rise. Pharmaceutical and biotechnology companies are investing heavily in advanced electronic data capture platforms, AI-enabled cleaning tools, and unified data warehouses to improve trial efficiency, reduce manual errors, and comply with stringent regulatory requirements set by authorities such as the FDA and EMA. The rapid shift toward decentralized and hybrid clinical trials is further accelerating demand for scalable cloud-based solutions that can integrate data from multiple sources, including wearables, ePRO systems, remote monitoring devices, and telehealth platforms.
Sponsors and CROs are also adopting real-time analytics dashboards that provide continuous oversight of patient safety, protocol adherence, and site performance, improving the speed of interim analyses and risk-based monitoring. Integration of electronic health records , genomic datasets, and imaging files into clinical workflows is driving the market toward more interoperable, AI-assisted systems. Overall, the industry is moving toward fully integrated, real-time clinical data management environments that enhance operational visibility, streamline regulatory submissions, and shorten the overall drug development timeline.
Market Trends
- Industry Growth Overview: The clinical data management industry is growing as clinical trials become more complex and data-heavy, driving increasing demand for cutting-edge digital platforms. Adoption of cloud-based AI-enabled solutions that improve speed and accuracy is accelerating due to increased R&D spending and the growth of decentralized trials.
- Sustainability Trends: Digital transformation is improving sustainability as businesses cut back on paper use, site visits, and manual labor. Automation and cloud systems are cutting waste and operational costs while supporting more efficient, environmentally responsible trials.
- Startup Ecosystem: New players in the startup ecosystem are providing quick, inexpensive AI-driven tools for data collection, validation, and decentralized trials. Competition is rising, and industry modernization is accelerating due to growing investment and innovation.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.62 Billion |
| Market Size in 2026 | USD 4.04 Billion |
| Market Size by 2035 | USD 10.89 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 11.64% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Type, Formulation, Application, End User, Distribution Channel, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Emerging Opportunities in the Clinical Data Management Market
|
Emerging Opportunity |
What it Enables |
Why it Matters |
|
AI & automation for data processing |
Automated data cleaning, validation, and anomaly detection |
Reduces workload, increases data accuracy, and shortens trial timelines |
|
Decentralized & hybrid trial platforms |
Remote patient data capture through mobile and wearables |
Expands patient reach, improves retention, and supports flexible trial models |
|
Real-world data (RWD) integration systems |
Integration of EHR, claims data, and patient-reported outcomes |
Strengthens clinical outcomes evidence and supports value-based approvals |
|
Cloud-based interoperable platforms |
Centralized data access with secure, scalable infrastructure |
Reduces IT costs, enables global collaboration, and supports multi-site trials |
|
Real-time analytics & performance dashboards |
Continuous tracking of trial status, patient safety, and compliance |
Enables faster decision-making and early detection of issues in ongoing trials |
|
Automated compliance & audit tools |
Standardized reporting, audit trails, and automated documentation |
Minimizes regulatory risk, reduces manual errors, and accelerates submissions |
|
Patient-centric engagement platforms |
Digital tools for consent, communication, and feedback |
Enhances patient experience and reduces dropout rates in long trials |
Clinical Data Management Market Segmental Insights
[[segment_insights]]
Clinical Data Management Market Regional Insights
[[regional_insights]]
Clinical Data Management Market Value Chain
[[value_chain]]
Clinical Data Management Market Companies
[[market_company]]
Recent Developments
- In August 2025, IQVIA and Veeva Systems announced a long-term global clinical and commercial partnership, along with the complete resolution of all pending legal disputes. The agreement allows customers to use software, data, technology, and services from both companies together, promoting seamless integration across platforms. (Source: https://www.iqvia.com )
- On 20 March 2025, Medidata Solutions unveiled its new Patient, Study, and Data Experiences during the NEXT London 2025 event, an AI-powered platform designed to unify clinical trial workflows, reduce costs, and accelerate therapy development by leveraging anonymized historical trial data. (Source: https://www.medidata.com )
Clinical Data Management Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
