September 2024
The global leukemia inhibitory factor (LIF) market size is calculated at USD 1.34 billion in 2025 and is forecasted to reach around USD 2.90 billion by 2034, accelerating at a CAGR of 8.94% from 2025 to 2034. The North America market size surpassed USD 500 million in 2024 and is expanding at a CAGR of 9.15% during the forecast period. The market sizing and forecasts are revenue-based (USD Million/Billion), with 2024 as the base year.
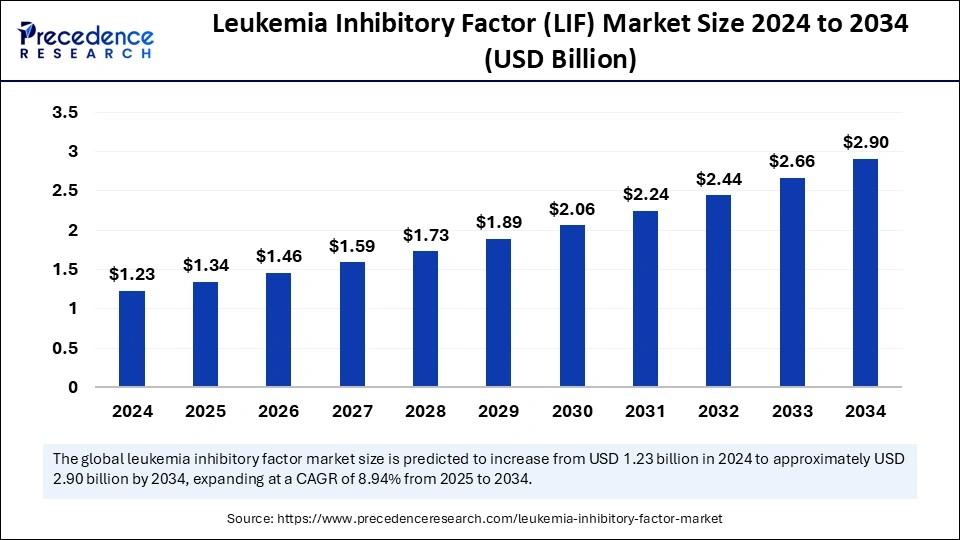
The global leukemia inhibitory factor (LIF) market size accounted for USD 1.23 billion in 2024 and is predicted to increase from USD 1.34 billion in 2025 to approximately USD 2.90 billion by 2034, expanding at a CAGR of 8.94% from 2025 to 2034. With the rising investments in cancer research and the development of novel cancer treatments, the leukemia inhibitory factor (LIF) market is experiencing exceptional growth globally.
Artificial intelligence technologies can analyze the complex roles of LIF in biological processes, stem cells, cancer, and immunity. AI can help researchers invent new therapeutic strategies by studying the roles of LIF in cell growth, cell differentiation, stem cell maintenance, and immune response. AI algorithms help in data analysis by analyzing datasets of LIF expression, cellular responses, and signaling pathways. AI can also predict cancer progression, identify new therapeutic targets, and develop personalized therapies.
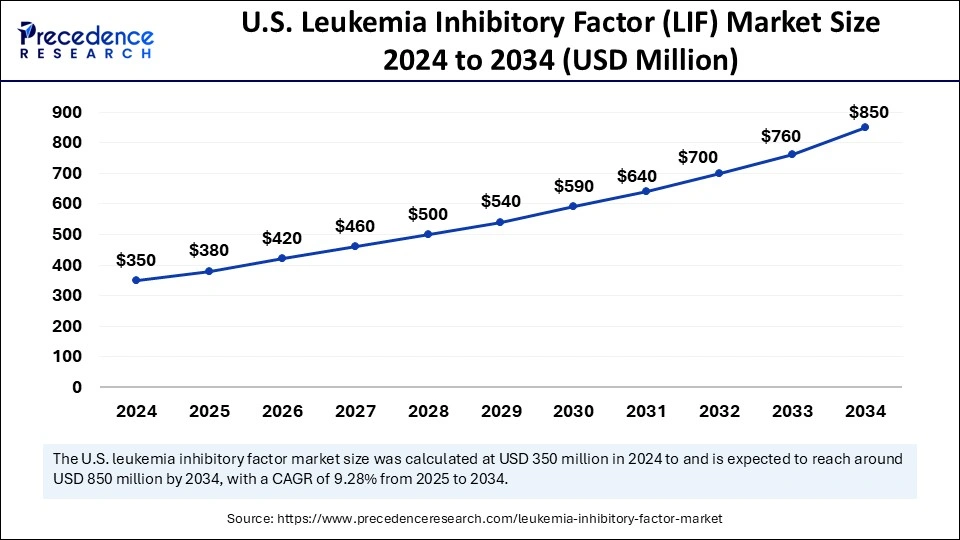
The U.S. leukemia inhibitory factor (lif) market size was exhibited at USD 350 million in 2024 and is projected to be worth around USD 850 million by 2034, growing at a CAGR of 9.28% from 2025 to 2034.
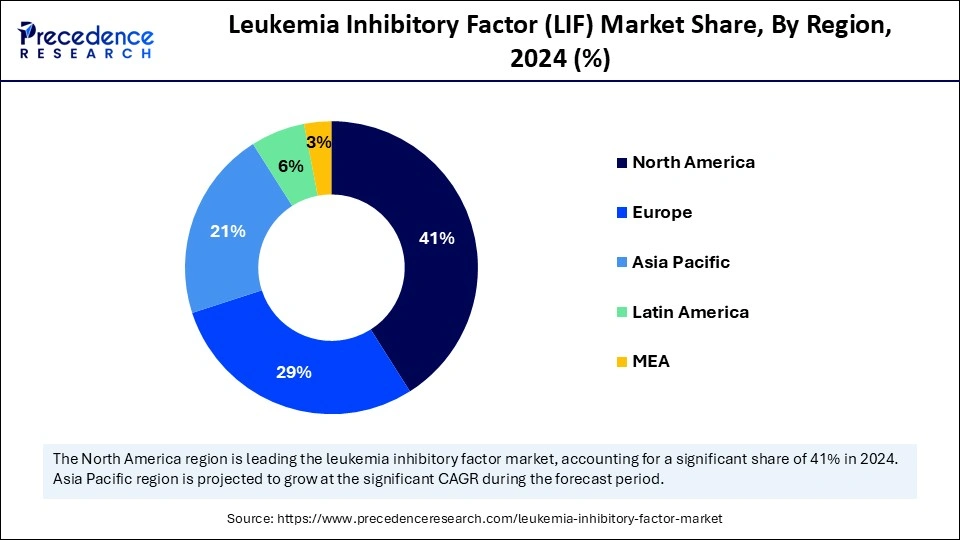
North America dominated the leukemia inhibitory factor (LIF) market in 2024 due to its well-established healthcare infrastructure, robust research and development activities, and high healthcare expenditures. The region also showcases a strong presence of the leading biotechnology companies, pharmaceutical companies, and research institutes.
The Leukemia & Lymphoma Society (LLS) reported that leukemia is the 10th most common cancer in the U.S. and that over 35.5 % more males than females are living with leukemia. The LLS also stated that chronic leukemia is expected to showcase 9.6% more cases than acute leukemia. The new cases of leukemia in older adults are diagnosed at the age of 67 years, which raises the need for research on LIF.
The doses of radiation, certain cancer therapies, and repeated exposure to the chemical benzene can cause acute myeloid leukemia (AML). Industrial emissions and automobile exhaust cause 20% of the total national benzene exposure. Furthermore, several cancer treatments, including immunotherapy such as CAR T-cell therapy and stem cell transplantation, raised the adoption of potential leukemia therapies among the patient population. The preference for tyrosine kinase inhibitors and such oral drugs to treat patients diagnosed with chronic myeloid leukemia (CML) also drives the expansion of healthcare facilities.
Rising Prevalence of Leukemia and Improved Treatments in the U.S.
The U.S. drives the market’s growth with extensive investments by companies and government entities for cancer research, biotechnology research, and stem cell research. About half of the benzene exposure is caused due to tobacco smoking, and exposure to tobacco smoke leads to acute myeloid leukemia (AML) among the U.S. population. The average exposure of smokers to benzene is 10 times daily than nonsmokers in the U.S. The emerging need for acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL) treatments immediately after diagnosis drives the market’s growth in the U.S. significantly. The various leukemia treatments, such as chemotherapy and targeted therapies including monoclonal antibody therapy, raise the significance of the leukemia inhibitory factor (LIF).
The Leukemia & Lymphoma Society (LLS) is located in New York with several regional offices in the U.S. and Canada. According to the facts 2023-2024 by the LLS, over 456,481 people are living with leukemia in the U.S. The LLS reported that 62,770 people are expected to be diagnosed with leukemia in 2024, while 23,670 people are expected to die from leukemia in 2024.
Asia Pacific is observed to host the fastest-growing leukemia inhibitory factor (LIF) market during the forecast period. The APAC regional progress is driven by rapid advancements in biomedical research and the emerging need for healthcare services. Countries such as India, China, and Japan contribute to significant investments in healthcare infrastructure and research capabilities. Government initiatives provide funds to promote stem cell research and regenerative medicine research. Furthermore, the rising incidence of neurological disorders and cancer has increased the growth of the LIF market.
The Asian Pacific Journal of Cancer Care (APJCC) is an open-access electronic journal that publishes papers related to clinical cancer research, focusing on the diagnosis, treatment, and prognosis of cancer patients. This APJCC was launched in the form of the official publication of the Asian Pacific Organization for Cancer Prevention (APOCP) by its West Asia Organization for Cancer Prevention (WAOCP).
Clinical Management of Leukemia Patients and the Healthcare System in Singapore
According to the Official Journal of the Academy of Medicine of Singapore, Singapore focuses on a balanced approach to therapy selection, side-effect profile, cost, integrating patient preferences, accessibility, and logistics for informed decision-making. Singapore accounts for 20 to 30 new cases of chronic lymphocytic leukemia (CLL) per year. This country provides treatments to patients at public or private institutions, including specialized hematology centers. The healthcare system of Singapore provides insurance, subsidies, self-pay, and co-pay options for patients. The drug’s local approval status determines the financing of outpatient cancer treatment in Singapore based on its inclusion in the Ministry of Health’s Cancer Drug List (CDL) and evaluations of its cost-effectiveness and clinical effectiveness. The subsidized access to drugs included in the CDL can be received by patients.
Europe is expected to be a significantly growing market for leukemia inhibitory factor (LIF) in the upcoming period. This growth is attributed to the robust R&D infrastructure and supportive regulatory framework. Countries such as France, the United Kingdom, etc., contribute to R&D activities and increase healthcare expenditure. The shift towards advancing stem cell research and cancer treatment drives the demand for LIF. Several biotechnology and pharmaceutical companies, along with research institutes, support scientific innovations and market growth.
Translational Research and Advancements in Spain
Spain accounts for several cancer cases among the population every year. With the rising prevalence of cancer, cancer treatments are also improving. Spain focuses on translational research, which allows the development of personalized treatment for cancer. Furthermore, genomics research offers improved ways to analyze the genetic profile of tumors along with their classification. Research in cellular biology studies tumorous cells and tissues and identifies the molecular basis of cellular behavior. Moreover, state-of-the-art technologies revolutionized faster diagnosis and efficient treatments. The Vall d'Hebron Institute of Oncology (VHIO), located in Barcelona, Spain, focuses on research related to preclinical and translational research, clinical research, and core technologies.
The leukemia inhibitory factor (LIF) market revolves around the extensive utilization of LIF for differentiating myeloid progenitor cells and inhibiting their growth. The efficient role of LIF in stem cell maintenance, stem cell regeneration, immune system regulation, as a cytokine, as a signaling molecule, etc., drives its adoption in biomedical and clinical research.
The major prevalence of four types of leukemia in the U.S. showcases acute lymphoblastic leukemia (ALL), chronic lymphocytic leukemia (CLL), acute myeloid leukemia (AML), and chronic myeloid leukemia (CML). The therapeutic potential of LIF in hematopoiesis, neuroprotection, and treating various conditions in the oncology and regenerative medicine sectors upholds its position in the market.
| Report Coverage | Details |
| Market Size by 2034 | USD 2.90 Billion |
| Market Size in 2025 | USD 1.34 Billion |
| Market Size in 2024 | USD 1.23 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 8.94% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Product Type, Application, End-User, and Regions. |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Biopharmaceutical Research and Biomedical Innovations
The rising prevalence of leukemia all around the world, especially in countries like the U.S., accelerates the growing investment plans and healthcare research. Advancements in biotechnology, pharmaceuticals, and clinical research promote the study of cytokines and their roles in cancer treatment to drive innovations. The government and private organizations provide funding to support the development of LIF-based therapies. The rising incidence of hematologic malignancies creates the need for advanced treatments and therapies that deliver precision and accuracy.
Inadequate Knowledge of Therapeutics
The LIF market faces some challenges related to the regulatory approvals for newly invented therapies. The clinical trial processes can be time-consuming and costly, which can create limitations to introducing new LIF-based treatments in the market. A lack of detailed understanding of the LIF mechanisms can create inconsistencies in research outcomes. A lack of awareness and incomplete understanding of the therapeutic potential of LIF among healthcare professionals can limit the market’s growth. The limited accessibility to advanced cancer treatments in some developing countries may present healthcare issues in front of the patient population.
Successful Clinical Trials and Testing of Pharmaceutical Drugs
The strategic collaborations between research organizations, academic institutions, and biopharmaceutical companies drive the innovative applications of LIF in treating various types of cancer. Scientific research focusing on drug discovery and development, oncology, etc., aims to deliver novel therapies to the market. The personalized medicine sector is creating ways to introduce targeted LIF therapies to fulfill patient needs by enhancing treatment efficacy and patient outcomes.
By product type, the recombinant LIF segment dominated the leukemia inhibitory factor (LIF) market in 2024 due to its widespread applications in research and therapeutics. The vast production of recombinant LIF through biotechnological methods drives its expansive reach in the marketplace. The wide utilization of the r-LIF in stem cell research helps to maintain pluripotency. Moreover, the utilization of the r-LIF for creating induced pluripotent stem cells (iPSCs) accelerates its demand among consumers.
By product type, the LIF antibodies segment is expected to grow rapidly in the leukemia inhibitory factor (LIF) market during the forecast period. This segmental growth is attributed to the importance of these components in biomedical research and clinical research. The high preference for LIF antibodies in the detection and quantification of LIF levels in several biological samples drives their huge adoption in research and healthcare. Scientists hugely prefer the LIF antibodies in cancer research to examine the role of LIF in tumor progression and metastasis. The growing shift towards cancer immunotherapy raised the demand for LIF antibodies by researchers to understand the immune response in cancer patients.
By application, the cancer treatment segment dominated the leukemia inhibitory factor (LIF) market in 2024 due to diverse applications of LIF in cancer treatment, neurological disorders, stem cell research, fertility treatment, etc. The potential ability of LIF to regulate cell growth and immune response made it a crucial target in the development of cancer therapies. The increased focus of researchers on LIF to innovate novel cancer treatments aims to optimize efficacy and eliminate side effects. The presence of various government organizations like the National Cancer Institute (NCI), the National Institute of Health (NIH), and the U.S. Food and Drug Administration (USFDA) provides strong support to promote cancer research and invent new cancer treatments globally.
By application, the stem cell research segment is anticipated to be the fastest-growing in the leukemia inhibitory factor (LIF) market over the forecast period. This remarkable growth is driven by the unique properties of LIF, such as the maintenance of the pluripotency of stem cells, which is crucial for regenerative medicine. The extensive utilization of LIF in research laboratories to culture embryonic stem cells and create induced pluripotent stem cells (iPSCs) that can differentiate into several cell types drives segmental growth. The importance of LIF for research in organ repair, regenerative therapies, and tissue engineering accelerates the overall expansion of stem cell research. The growing shift toward stem cell-based treatments raises the demand for LIF in the market.
By end user, the hospitals segment dominated the leukemia inhibitory factor (LIF) market in 2024 due to the presence of several hospitals as the principal end-users of LIF-related services. The adoption of LIF by hospitals for cancer treatment and fertility services also raises segmental growth in the market. The employment of LIF-based therapies and diagnostic tools by hospitals offers advanced treatment options to patients. The insertion of LIF into clinical research practices, oncology research, and reproductive medicine drives segmental growth remarkably.
By end user, the research institutes segment is observed to grow at the fastest rate in the leukemia inhibitory factor (LIF) market during the forecast period. Research institutes stand as the vital end-users of LIF-based services. The efforts to study the biological functions and therapeutic potential of LIF drive the expansive success of research institutes all over the world. Research conducted on the role of LIF in cellular processes, therapeutic applications, and disease mechanisms accelerates the growth of academic and government research institutes. The increasing investments in biomedical research favor novel therapeutic innovations.

By Product Type
By Application
By End-User
By Geography
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
September 2024
February 2025
August 2024
August 2024