February 2025
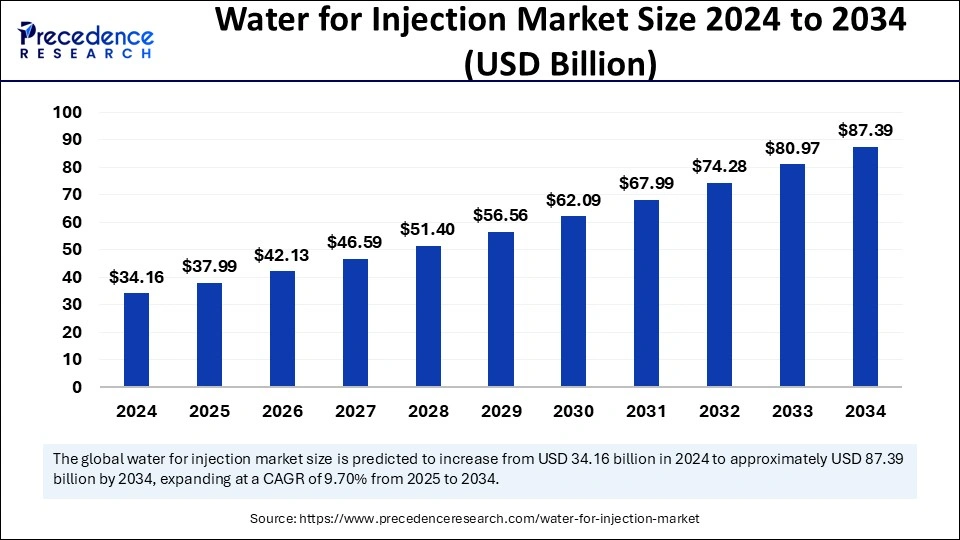
The global water for injection market size is calculated at USD 37.99 billion in 2025 and is forecasted to reach around USD 87.39 billion by 2034, accelerating at a CAGR of 9.70% from 2025 to 2034. The North America market size surpassed USD 13.57 billion in 2024 and is expanding at a CAGR of 9.78% during the forecast period. The market sizing and forecasts are revenue-based (USD Million/Billion), with 2024 as the base year.
The global water for injection market size was estimated at USD 34.16 billion in 2024 and is predicted to increase from USD 37.99 billion in 2025 to approximately USD 87.39 billion by 2034, expanding at a CAGR of 9.70% from 2025 to 2034. The demand for injectable medications and biologics, as well as biosimilars, has increased, driving the need for high-quality water for injections.
The water for injection market production and management processes have witnessed a significant favorable impact with the integration of Artificial Intelligence. AI algorithms optimize processes, improve predictive maintenance, monitor water quality, reduce energy consumption, and need a more cost-effective solution. They aid in identifying potential areas for improvement. The real-time monitoring and data-driven decision-making capabilities of AI are key to transforming water quality. Machine learning (ML) algorithms provide large data analysis, identify patterns, and offer predictions to make data-driven decisions and improve water injection operations. Pharmaceutical manufacturers are rapidly adopting AI implementations to meet stringent pharmaceutical regulatory standards and improve the quality of water for injection.
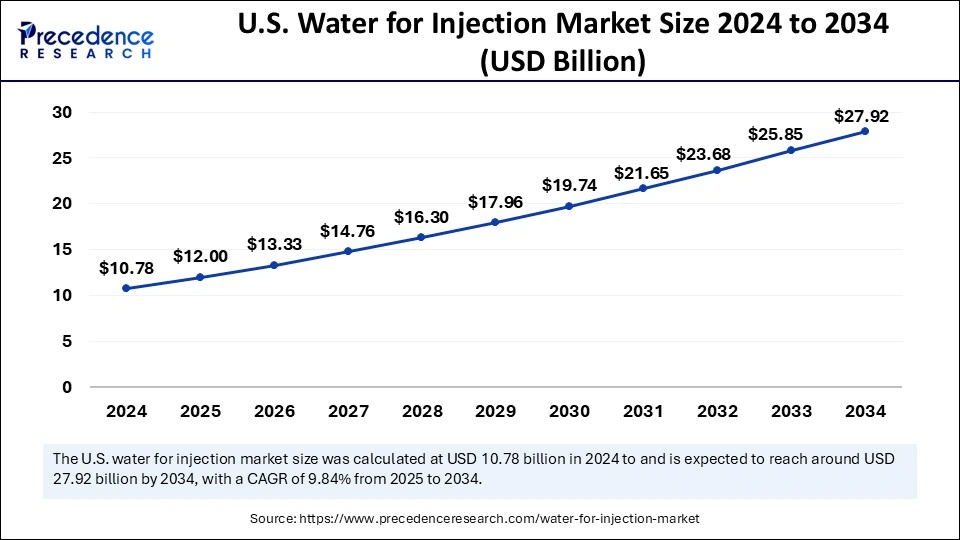
The U.S. water for injection market size was exhibited at USD 10.78 billion in 2024 and is projected to be worth around USD 27.92 billion by 2034, growing at a CAGR of 9.84% from 2025 to 2034.
Stringent Regulatory Environments: To Boost North American Market
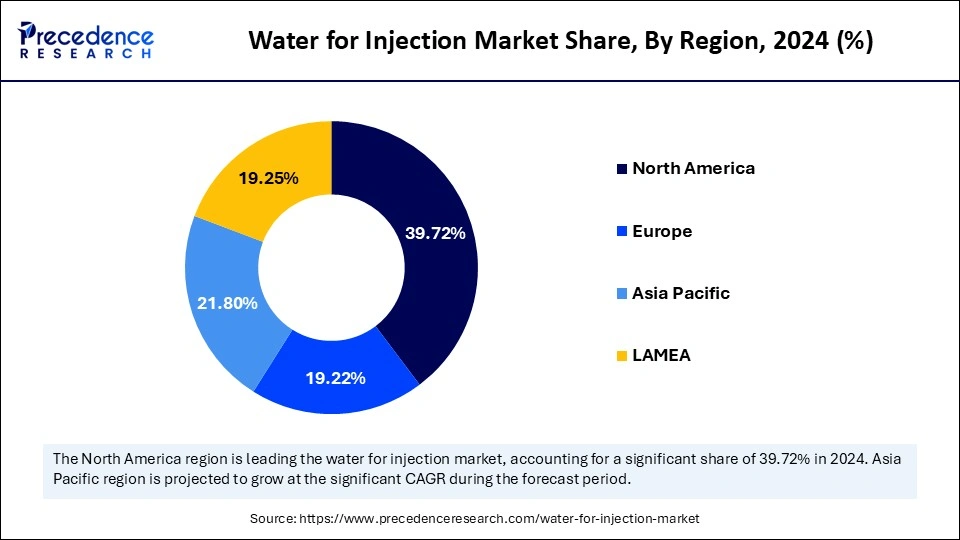
North America dominates the global water for injection market due to various factors like the large pharmaceutical industry, advanced manufacturing infrastructure, high demand for parenteral drugs, and the presence of a strong regulatory environment. North America has witnessed high demand for injectable medications and biologics fuels the need for water for injection products. Advanced manufacturing infrastructure and key vendors of the region are focusing on enabling the production of high-quality water for injections. Additionally, ongoing developments of cost-effective water for injection production methods and the adoption of innovative technologies like energy-efficient distillation and reverse osmosis systems are expanding the market.
The large pharmaceutical and biotechnology industries of the United States are high contributors to the water for injection market. The stringent regulatory standards drive the requirement of Distillation, including multi-effect and vapor compression distillation, for water for injections. The requirement of complying with regulatory compliance with ultra-pure water drives spectacular approaches in advancements in pharmaceutical water production. The United States is the prime adopter of Reverse osmosis systems. The United States is expected to continuously dominate the market with its advanced healthcare sector and the existence of key market players.
Asia Pacific Market Rising with Expanding Pharmaceutical and Healthcare Sector
Asia Pacific will host the fastest growing water for injection market during the forecast period. The expanding pharmaceutical industry, the presence of key market players, and increased demand for injectable medications are the factors contributing to this growth. Asia Pacific is a provider of low-cost manufacturing options, making it an ideal location for water injection production. Improving pharmaceutical capabilities and international collaborations empowering the advancements in water for injection productions. Regional improvements in regulatory standards are driving demand for ultra-pure water in injectable drug production.
Countries like India, China, and Japan are leading the Asia Pacific water for injection market due to higher spending on healthcare, increased government initiatives and support for research and development, and a focus on clinical trials. India is expected to hold a significant market share in the upcoming period due to the rapidly growing pharmaceutical industry and demand for injectable medications. Government funding and investments in the pharmaceutical industry contribute to expanding the market.
Water for injection (WFI) is a crucial component in the manufacturing industry, providing high-quality water for the production of pharmaceutical ingredients, injectable medications, and drug formulations. The water for injection market has witnessed transforming growth due to increased research activities, clinical trials, and pharmaceutical manufacturing procedures, driven by an increase in demand for injectable medications such as vaccines, insulin, and monoclonal antibodies. The demand for biopharmaceuticals, biosimilars, and biologics is further contributing to this growth.
The increased focus on patient safety has driven pharmaceutical manufacturers' emphasis on advancing high-quality water for injection procedures. Technological advancements such as vapor compression, multiple effect distillation, reverse osmosis systems with a downstream unit of ultrafiltration, and membrane-based reverse osmosis are improving the quality and efficiency of water for injection productions. The availability of more efficient and cost-effective water for injection production methods approaches spectacular advantages in pharmaceutical manufacturing.
| Report Coverage | Details |
| Market Size by 2034 | USD 87.39 Billion |
| Market Size in 2025 | USD 37.99 Billion |
| Market Size in 2024 | USD 34.16 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 9.70% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Application, End-User, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
The trend of single-use systems
Pharmaceutical companies are rapidly adopting single-use systems to reduce the risk of contamination and increase efficiency. The single-use system promotes significant advancements of sustainability in the pharmaceutical manufacturing of therapeutic drugs and equipment, like chemicals and resources, like water and energy, needed to sterilize reusable systems. The single-use system reduces costs associated with cleaning, validation, and maintenance. Water for injection is the crucial compound to support the reduction of production costs by using a single-use system. The adoption of high-quality water for equipment cleaning and media production has increased due to the extreme use of single-use systems.
High production cost
Distillation and reverse osmosis are major processes in the water for injection market. The distillation process requires a significant amount of energy. The distillation and reverse osmosis process requires expensive equipment and maintenance for the production of water for injections. The production of water for injection requires meeting strength quality control standards. Additionally, the need for a pharmaceutical manufacturer to comply with regulatory requirements further contributes to high costs. However, technological advancements such as vapor compression distillers are significant approaches in the reduction of operation noise levels, energy intensity, and maintenance.
Advancements in membrane-based reverse osmosis systems
Technological advancements in the water for injection market, such as the improvement of strain quality and advancements in membrane-based reverse osmosis systems, are providing high-quality water solutions. The need to meet the strong regulatory requirements of water for injection is driving the adoption of membrane-based reverse osmosis systems in pharmaceutical manufacturing. This system reduces the risk of contamination in production and monitors potential needs.
The membrane-based reverse osmosis system takes less space and is cost-effective compared to distillers. This system requires low maintenance, reduces downtime, and increases overall reliability. The flexibility and scalability of the system in water for injection production make it an attractive option for pharmaceutical manufacturing. Manufacturers are focusing on advancing systems to enable efficient water production.
The formulated parental drugs segment dominated the water for injection market in 2024. The increased demand for parenteral drugs like vaccines, insulin, and monoclonal antibodies is driving demand for water for injections. Water for injection is crucial for preserving the integrity and potency of parenteral drugs, as it is a critical component of parenteral drugs, used as solvents and diluents for pharmaceutical ingredient activities. The formulation of parenteral drugs demands high-quality water for injections. Advancements in distillation and membrane-based purification technologies to improve the efficiency of water for injections are increasing their popularity in the formulation of parenteral drugs. Additionally, the increased demand for biopharmaceuticals, biosimilars, and biologics drives the need for water for injections in formulations of parental drugs.
The solvent segment is expected to grow at the fastest CAGR over the forecast period due to the increased utilization of solvents in pharmaceutical manufacturing. Water for injections is used as a solvent in the synthesis of active pharmaceutical ingredients (APIs), drug formulations, and the cleaning of equipment in pharmaceutical manufacturing facilities. The stringent regulatory requirements for high-quality safety standards and highly scalable solvents are driving the utilization of water for injections in the synthesis process. Synthesis solvents are crucial in improving the efficiency of pharmaceutical manufacturing.
Under the solvent segment, the synthesis of drugs sub-segment holds a significant share, leading the market with a growing demand for injectable medications. Water for injection is crucial for the formulation of drugs as it is used to dissolve and stabilize the active pharmaceutical ingredients. It is a primary solvent in the production of parenteral drugs, which drives a high demand for water for injections. The use of high-quality water as a solvent in drugs, reconstitution and synthesis, vaccine and biologics manufacturing, and ensuring the safety and efficacy of medications drive the need for large quantities of water for injections.
Water for Injection Market Revenue, By Application, 2022-2024 (USD Billion)
| Application | 2022 | 2023 | 2024 |
| Formulate Parenteral Drugs | 17.80 | 19.91 | 22.21 |
| Solvent | 5.64 | 6.31 | 7.04 |
| Cleaning Agents | 3.95 | 4.41 | 4.91 |
The pharmaceutical and biotechnology companies segment led the water for injection market in 2024. Pharmaceutical and biotechnology companies are the prior adapters of water for injections due to their high need for drug formulation, equipment cleaning, and quality control. Pharmaceutical and biotechnology companies have large-scale production for injectable medications. The growing demand for biologics and biosimilars is a driving need for water for injections in these companies. Increase focus on patient safety by driving demand for high-quality water for injections to reduce the risk of contamination and uninsured the quality and safety of injectable medications.
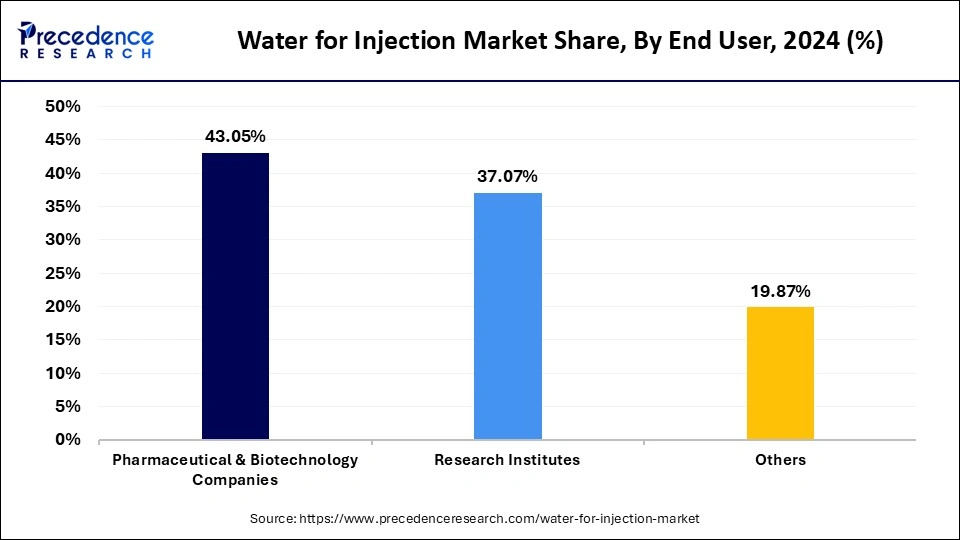
The research institutes segment is anticipated to witness significant growth in the forecast period. The adoption of water for injections has increased in research institutes due to increased R&D activities and clinical trials. Research institutes have witnessed high demand for high-quality water for injections for the development of novel pharmaceuticals and biologics. The requirement to adhere to strong quality control regulations to ensure the accuracy and reliability of research further contributes to the increasing demand for high-quality water for injections.
Water for Injection Market Revenue, By End-User, 2022-2024 (USD Billion)
| End User | 2022 | 2023 | 2024 |
| Pharmaceutical & Biotechnology Companies | 11.78 | 13.18 | 14.71 |
| Research Institutes | 10.15 | 11.35 | 12.66 |
| Others | 5.46 | 6.10 | 6.79 |
By Application
By End-user
By Geography
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
February 2025
January 2025
January 2025
February 2025