What is the Drug Device Combination Products Market Size?
The global drug device combination products market size is calculated at USD 165.26 billion in 2025 and is predicted to increase from USD 180.18 billion in 2026 to approximately USD 382.67 billion by 2035, expanding at a CAGR of 8.76% from 2026 to 2035. The need for combination products that are easy to use is increased by the trend towards home-based healthcare services, particularly in the aftermath of the COVID-19 epidemic.
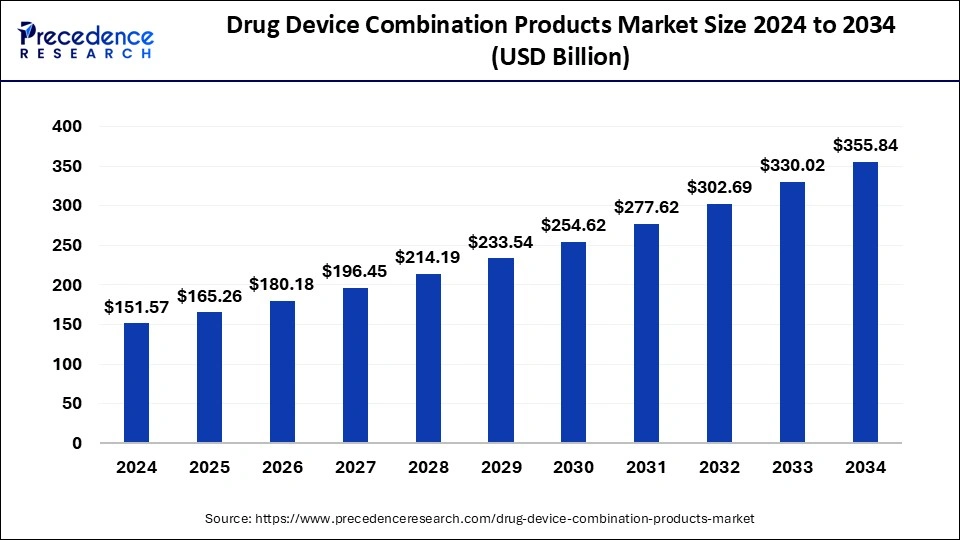
Drug Device Combination Products Market Key Takeaways
- The global drug device combination products market was valued at USD 151.57 billion in 2025.
- It is projected to reach USD 355.84 billion by 2035.
- The drug device combination products market is expected to grow at a CAGR of 8.91% from 2026 to 2035.
- North America held the largest share of the drug device combination products market in 2025.
- Asia Pacific is expected to host the fastest-growing market during the projected period.
- By product, the transdermal delivery system segment led the global market in 2025.
- By product, the inhaler segment is expected to grow at a significant rate during the forecast period.
- By application, the respiratory problem segment dominated the market in 2025.
- By application, the antimicrobial segment is expected to grow rapidly in the market in the upcoming years.
- By end use, the hospitals & clinics segment held the largest share of the market in 2025.
- By end use, the ambulatory surgical center segment is expected to grow at a notable rate during the forecast period.
What is a Drug Device Combination Products?
Therapeutic and diagnostic products that mix pharmaceuticals, devices, and/or biological components are known as drug-device combination products. Because these products can improve therapy efficacy and safety, their popularity is growing. They are frequently used to treat conditions like diabetes, respiratory issues, cardiovascular illness, and more. An increase in the prevalence of respiratory illnesses, cardiovascular ailments, and diabetes
Stents are frequently used in cardiovascular conditions and release medication to stop artery blockage. The largest drug device combination products market is a result of a vast patient base, significant healthcare spending, and sophisticated healthcare infrastructure. Growth in customized drug-device combo products that are made to meet the demands of each patient individually.
The drug device combination products market is expected to develop significantly due to factors like aging populations, increased chronic disease prevalence, and technological advancements. Even though there are obstacles in the way, continued innovation and supportive regulations should drive the sector forward. To address the changing demands of the healthcare industry, major firms are concentrating on creating cutting-edge, customized solutions.
IoT and AI integration for more intelligent and effective medication delivery systems. Accelerating the creation of wearable technology to give drugs continuously. The drug device combination products market access may be impacted by regional variations in reimbursement laws. The fastest-growing region is being driven by population growth, rising healthcare spending, and expanding awareness of cutting-edge medical treatments.
Artificial Intelligence: The Next Growth Catalyst in Drug Device Combination Products
AI is profoundly reshaping the drug-device combination products industry by enabling the development of smart delivery systems that enhance patient adherence and optimize treatment outcomes, such as AIenabled insulin pumps and advanced inhalers. By analyzing vast patient datasets, AI allows for personalized medicine, tailoring dosage and release profiles to individual patient needs in real time.
In the development phase, machine learning accelerates the identification of optimal drug formulations, reducing the reliance on costly, time-consuming trial-and-error laboratory methods.
What are the Growth Factors in the Drug Device Combination Products Market?
- One key motivator is the need for less intrusive procedures. Benefits like less pain, less time spent in the hospital, faster recuperation periods, and increased safety and efficacy make these procedures more and more appealing to patients and medical professionals.
- Chronic illnesses like cancer, diabetes, and cardiovascular problems are becoming more and more common. Because of this, there is a growing need for cutting-edge combination products that can improve therapy results. For example, insulin pumps and drug-eluting stents are becoming necessary for the proper management of certain disorders.
- New combination products are finding it easier to get approved and used, especially in North America, where there are strong regulatory frameworks and policies in place. Manufacturers are able to introduce new items onto the market more quickly because of streamlined approval procedures and unambiguous safety rules.
- Favorable conditions for the drug device combination products market expansion are being created by rising healthcare spending and bettering healthcare infrastructure, particularly in developing nations like China and India. In these areas, the use of combination products and cutting-edge medical treatments is becoming more widespread.
- Pharmaceutical companies, medical device makers, and academic institutions are collaborating strategically to accelerate the development and commercialization of novel combination medicines. These collaborations are essential for utilizing complementary knowledge and assets.
- The need for combination products that address age-related health conditions is being driven by the aging population, especially in North America, Europe, and Asia Pacific. The changing demographics are causing a rise in the demand for cutting-edge medical procedures and equipment.
Drug Device Combination Products Market Outlook
- Industry Growth Overview: The market is undergoing robust growth, driven by aging populations, chronic disease prevalence (diabetes, cardio), need for patient-centric care (home use, self-administration), and even tech advancements (smart devices, precise delivery).
- Major Investors: It includes large MedTech and Pharma giants such as Medtronic, Abbott, Boston Scientific, Stryker, Becton Dickinson (BD), Novo Nordisk, Novartis, and Eli Lilly, who invest depends on R&D, strategic acquisitions, and even product innovation, focusing on areas such as smart insulin pens, infusion pumps, drug-eluting stents, and connected inhalers, driving market expansion alongside private equity and government supporting for research.
Market Outlook
- Market Growth Overview: The drug device combination products market is expected to grow significantly between 2025 and 2034, driven by the shift towards home care and self-administration, high demand for drug-eluting stents, infusion pumps, and inhalers for cancer and cardiovascular diseases, and innovation in targeted, localized, and sustainable drug release technologies.
- Sustainability Trends: Sustainability trends involve eco-friendly materials, at-home care, digital health, and design for sustainability.
- Major Investors: Major investors in the market include Abbott, Medtronic, Novartis AG, Novo Nordisk A/S, Terum Corporation, and Vanguard.
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 382.67 Billion |
| Market Size by 2026 | USD 180.18 Billion |
| Market Size in 2025 | USD 165.26 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 8.76% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Application, Product, End-use, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Personalized medicine
Precision medicine, another name for personalized medicine, adjusts medical care to each patient's unique needs. This strategy is revolutionizing the market for drug-device combo goods by combining tailored medicines with cutting-edge diagnostic technologies. Finding biomarkers is critical to personalized medicine. These biomarkers are found by diagnostic technologies, which makes it possible to tailor medication regimens.
Companion diagnostics can assess a patient's likelihood of benefiting from a specific targeted medication. Treatment regimens can be modified in real-time thanks to the constant patient data collection provided by wearable technology and mobile health applications. Treatments are dynamically adapted to the patient's present state according to this data-driven method.
Restraint
Supply chain vulnerabilities
There are multiple supply chain vulnerabilities in the drug device combination products market. Goods include products like transdermal patches, pre-filled syringes, inhalers, and drug-coated stents. These weaknesses may have a major effect on these essential healthcare goods' costs, quality, and availability. It can be difficult and time-consuming to ensure compliance with rules regarding both drugs and medical devices, which could cause delays.
Products must adhere to the regulatory standards set forth by the agencies that oversee medical devices and pharmaceuticals, which may differ greatly. Getting regulatory body permission can be a drawn-out procedure that causes delays in product release. Quick technical progress can make current items outdated, necessitating ongoing investment in new technology.
Opportunity
Digital health integration
A major step forward in healthcare is represented by digital health integration in the drug device combination products market, which aims to enhance patient outcomes, adherence, and overall healthcare efficiency. Digital health technology (such as Bluetooth, sensors, and mobile apps) can be integrated into these items to improve their functionality.
Sensors can track consumption and dose, giving patients and medical professionals access to real-time data. By tracking adherence, informing patients of possible problems, and serving as a reminder for medicine take-home, digital health technologies can increase patient adherence rates. With easy access to their health data and real-time feedback, patients take a more active role in their own care.
Segment Insights
[[segment_insights]]
Regional Insights
[[regional_insights]]
Value Chain Analysis of Drug Device Combination Products Market
- R&D
This integrates pharmaceutical and device development, targeting on seamless drug-device compatibility, user-centric design (such as auto-injectors, inhalers, stents), Quality by Design (QbD), integrated clinical/regulatory strategies, and risk management, requiring close partnership between device engineering, drug formulation, and regulatory teams to ensure stability, safety, dosage accuracy, and also performance throughout the product's lifecycle. - Packaging and Serialization
It includes a dual regulatory framework, integrating demands for both pharmaceuticals and medical devices. The main goal is to guarantee end-to-end traceability throughout the supply chain to thus,combat counterfeiting, manage recalls, and ensure patient safety.
Drug Device Combination Products Market Companies

- Medtronic: Medtronic provides for the drug-device combination products market because it addresses huge expansion drivers such as rising chronic diseases, need for personalized/painless delivery, and even tech advancements, utilizing its strong R&D, broad portfolio (stents, pumps, pens), and global reach, along with strategic acquisitions to dominate with some integrated solutions for conditions such as cardiovascular disease, pain, and diabetes, working with patient requirements for better adherence and outcomes.
- Abbott Laboratories: Abbott Laboratories provides drug-device combinations in the growing market as these products offer superior patient results (targeted delivery, better compliance) for chronic disorders (cardio, diabetes), meet demand for integrated solutions, leverage Abbott's strong device portfolio (stents, infusion pumps), and provide cost-efficiency for healthcare systems, positioning them as a leader with developed tech such as drug-eluting stents.
Other Major Key Players
- Boston Scientific Corporation
Recent Developments
- In January 2026, BD and Ypsomed announced the development of a 5.5 mL version of the BD Neopak™ XtraFlow™ Glass Prefillable Syringe, specifically integrated with the YpsoMate 5.5 autoinjector. The product is designed to facilitate large-volume subcutaneous self-injections for high-dose therapies. (Source: https://investors.bd.com )
- In January 2024, the pharmaceutical, biopharmaceutical, and medical device industries can now access integrated and stand-alone analytical support from Kindeva Drug Delivery, a global supplier of drug device combination products, as part of its expanded analytical services capabilities. Kindeva Drug Delivery recently launched a new global business unit. Over the course of more than 50 years, Kindeva has gained a great deal of knowledge, experience, and competence while working on cGMP commercial supply and drug delivery development programs for transdermal, injectable, and inhalation routes.
- In July 2023, the first generic version of AstraZeneca's Symbicort, BreynaTM (budesonide and formoterol fumarate dihydrate) Inhalation Aerosol, has been approved by the U.S. Food and Drug Administration (FDA) and is the first product of a collaboration between Kindeva Drug Delivery L.P. and Viatris Inc. a global healthcare company. Breyna is a medication-device combination product that will be made available right away in dosage levels of 80 mcg/4.5 mcg and 160 mcg/4.5 mcg. It is recommended for specific people who suffer from asthma or chronic obstructive pulmonary disease (COPD).
Segment Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
